
Thin film fabrication techniques can be categorized into two procedures representing dry and wet processes. The chapter of “Methods of Fabricating Thin Films for Energy Materials and Devices” describes the overview of the methodology for thin film fabrication. This book includes four categories: (1) method for thin film fabrication, (2) cathode, (3) anode, and (4) solid electrolyte, written by world authorities in these fields. The optimal combination of these materials can yield a battery that is light, thin, long-lasting, and safe. The optimal electrolyte should be an efficient ion-conductor and a good electrical insulator allowing the battery to operate safely. The current research of electrolyte, whose form is preferable to be solid in thin film batteries, trends toward ceramics such as lithium lanthanum zinc oxide (LLZO) and lithium lanthanum titanium oxide (LLTO). Both the cathode and anode materials are film, chosen for their ability to intercalate, and de-intercalate lithium ion while maintaining their structural integrity. Anode materials are lithium metal, carbon-based materials, and inorganic compounds. Cathode materials are often mixed metal oxides involving lithium ion such as LiCoO 2 and LiMn 2O 4. As the demands for safety, higher energy density, and other performance metrics increase, research into anode, cathode, and electrolyte materials has been rapidly progressing. The thin film LIB is composed of the anode, cathode, and electrolyte with thicknesses on the order of microns. Thin film LIB is one of the forms of LIB. It has attracted much interest for use as power sources of smart cards, implantable medical devices, micro-sensors, and so on. Many current types of research for LIB focus on life extension, energy density, safety, cost reduction, and charging speed. Lithium-ion batteries are evolving even now. The LIB has revolutionized our lives and is widespread from small-scale devices such as mobile phone to emergency distributed power supply, electric vehicle, etc. The energy density of LIB with high discharge voltage (3.6 V) is nearly twice that of Ni-Cd batteries, and excellent cycle life and higher level of intrinsic safety have been demonstrated. 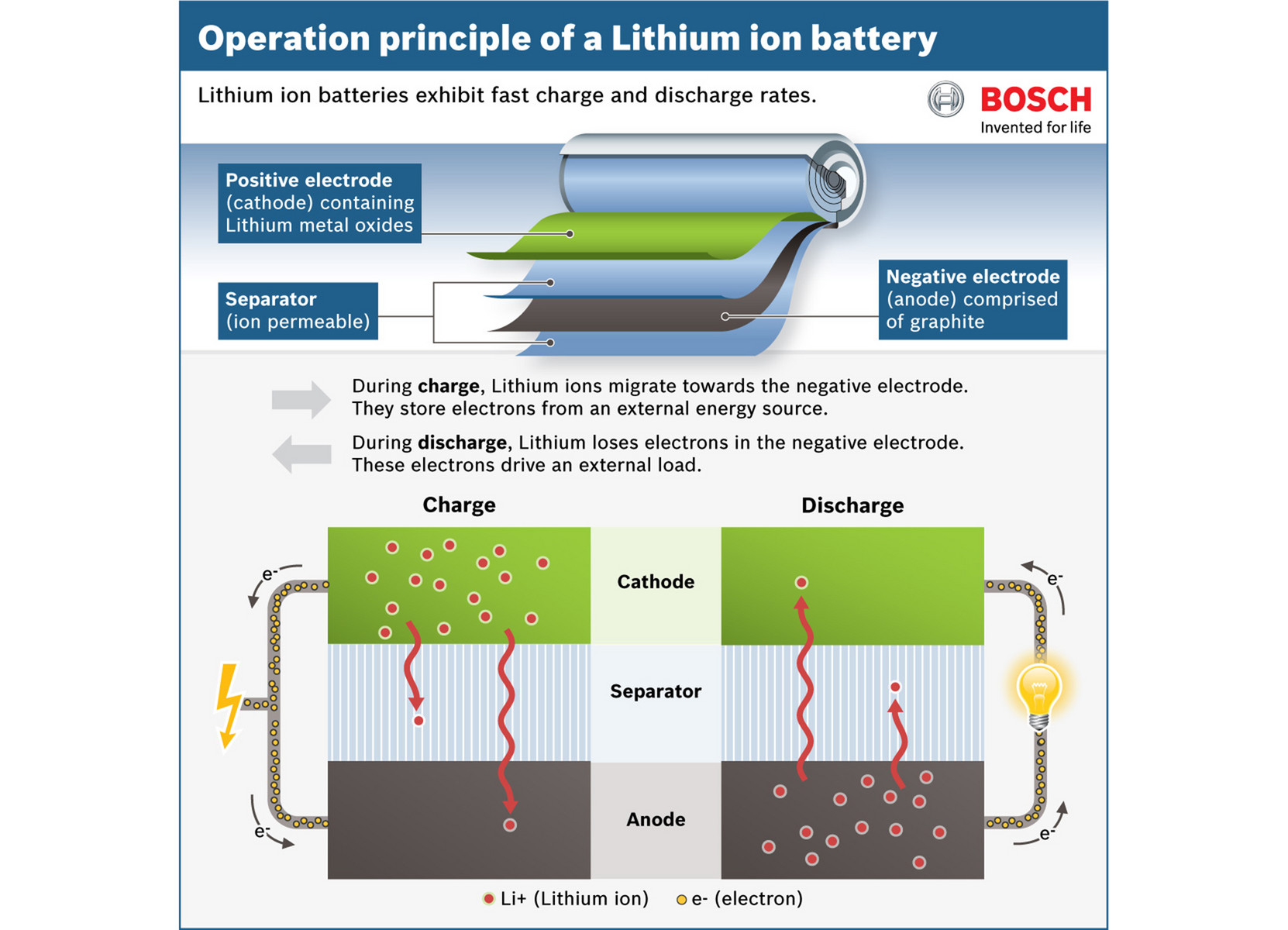
It is the invention of lithium-ion battery (LIB). In addition, the accuracies and computational efforts of the four equivalent circuit models are compared.In 2019, the Nobel Prize in Chemistry has been awarded to John B. Goodenough, M. Stanley Whittingham, and Akira Yoshino for their research in improving battery technology. Therefore, as a result, this study presents a concise but comprehensive battery parameter library that should aid battery system designers or power electronic engineers in conducting battery simulations and in selecting appropriate battery cells based on application-specific requirements. For example, it is shown that the energy density of modern 18650 cells can vary from a typical value of 200 to about 260 Wh kg −1, whereas the cell price can deviate by a factor of about 3 to 5. Furthermore, the pricing, impedance, specific energy, and C-rate of the chosen battery cells are compared. Their impedance properties, according to four different equivalent circuit models, are measured using electrochemical impedance spectroscopies. Various commercially available cylindrical, state-of-the-art lithium-ion battery cells, both protected and unprotected, are considered. Electrical equivalent circuit models, either linear or nonlinear, are commonly used for this purpose and are presented in this article. Electrical models of battery cells are used in simulations to represent batteries’ behavior in various fields of research and development involving battery cells and systems.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
